Imatinib Side Effect and Safety
SEO Meta & Title
Page Title
Imatinib Side Effect and Safety
Meta Description
Learn about Imatinib side effect and safety, common risks, serious warnings, generic options, pricing, and trusted India supply solutions in 2026.
Imatinib Side Effect and Safety: Complete 2026 Guide for Patients and Buyers
Understand Imatinib side effect and safety before starting treatment. Explore clinical safety data, common side effects, India generic options, and verified procurement support from 9S Pharmacy Service.
CTA Buttons
Talk to Oncology Supply Expert
Suggested Hero Image
Drug Overview
Imatinib is one of the most important targeted cancer therapies introduced in modern oncology. It is widely prescribed for chronic myeloid leukemia (CML), gastrointestinal stromal tumors (GIST), Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL), and several rare hematologic conditions.
Originally developed and marketed as Gleevec by Novartis, Imatinib changed cancer treatment by specifically targeting abnormal tyrosine kinase activity instead of using only conventional chemotherapy. It was first approved by the U.S. FDA in 2001 and rapidly became a global standard of care. WHO and NCCN treatment guidelines continue to recommend Imatinib as a foundational therapy for several indications.
The mechanism of action focuses on inhibiting BCR-ABL tyrosine kinase, the abnormal protein responsible for CML progression. It also blocks KIT and PDGF receptor signaling pathways involved in GIST and related tumors. This targeted inhibition slows or stops cancer cell growth while reducing damage to healthy cells compared to older chemotherapy methods.
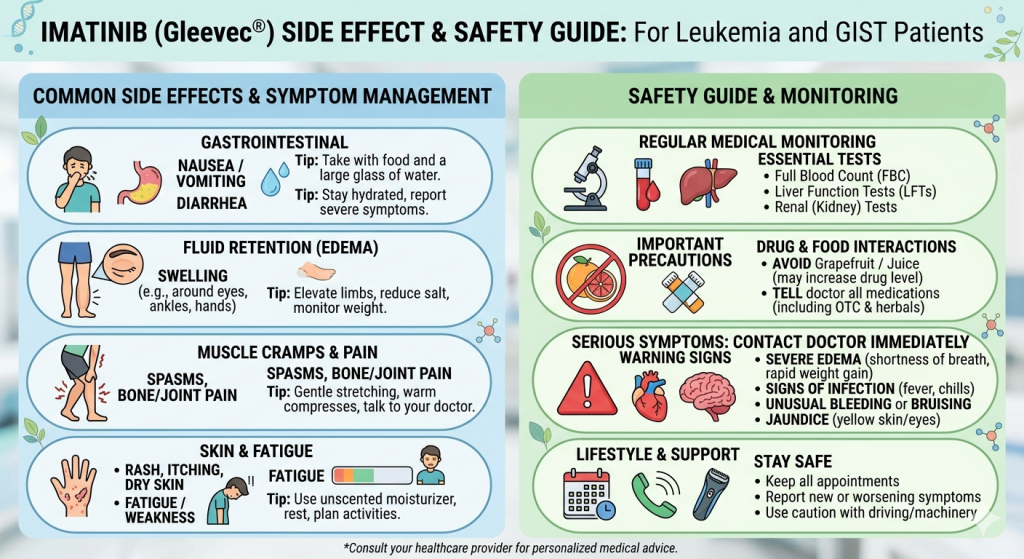
When discussing Imatinib side effect and safety, patients most commonly experience nausea, fatigue, edema, muscle cramps, diarrhea, rash, and mild liver enzyme elevation. Serious but less common risks include hepatotoxicity, severe fluid retention, cardiac effects, bone marrow suppression, and rare gastrointestinal bleeding. Regular blood monitoring is essential during therapy.
Clinical studies showed excellent long-term survival benefits. The IRIS trial demonstrated major survival improvements for CML patients using Imatinib, with many patients achieving durable remission beyond 10 years. These outcomes helped establish the drug as one of the most successful targeted oncology therapies globally.
India now supplies high-quality WHO-GMP and USFDA-compliant generic Imatinib through leading manufacturers including Cipla, Natco, Hetero, Sun Pharma, and Dr. Reddy’s. These versions significantly reduce treatment cost while maintaining therapeutic equivalence.
Original Brand vs India Generic Comparison
| Parameter | Original Brand (Glivec) | India Generic Imatinib |
|---|---|---|
| Manufacturer | Novartis | Cipla / Natco / Hetero / Sun Pharma |
| Main Strength | 100mg / 400mg | 100mg / 400mg |
| Monthly Cost | Very High | 60–85% Lower |
| Regulatory Standard | USFDA / EMA | WHO-GMP / USFDA / EU GMP |
| Global Supply Access | Limited by pricing | Broad international export support |
Why Choose India Generic
India has become the world’s leading supplier of oncology generics, especially for long-term therapies like Imatinib. Buyers looking for better affordability without compromising quality increasingly prefer India generic options.
- Cost Savings: Generic Imatinib is commonly 60–85% less expensive than the original brand.
- WHO-GMP Certified Facilities: Major manufacturers follow strict global compliance standards.
- USFDA and EU GMP Approvals: Many facilities are internationally audited and export-ready.
- Large Production Capacity: High-volume manufacturing supports stable global supply.
- Reliable API Ecosystem: India has strong domestic active pharmaceutical ingredient production.
- 9S Network Assurance: Verified sourcing reduces procurement risks and improves documentation transparency.
- Flexible MOQ Options: Suitable for hospitals, distributors, NGOs, and procurement agencies.
- Faster Export Support: Strong logistics network improves lead times for international delivery.
According to global pharmaceutical export reports and WHO manufacturing assessments, India continues to lead the oncology generic sector in both production scale and regulatory inspection frequency. This makes generic Imatinib a practical and trusted option for both patients and institutional buyers.
Member Manufacturers Directory
| Company | Strengths Available | Monthly Capacity | MOQ | Certification | Action |
|---|---|---|---|---|---|
| Cipla | 100mg / 400mg | 1M+ boxes | 100 boxes | WHO-GMP, USFDA | Request Quote |
| Natco Pharma | 100mg / 400mg | 800K+ boxes | 100 boxes | WHO-GMP, EU GMP | Request Quote |
| Hetero | 100mg / 400mg | 900K+ boxes | 100 boxes | WHO-GMP, USFDA | Request Quote |
| Sun Pharma | 100mg / 400mg | 1.2M+ boxes | 100 boxes | WHO-GMP, USFDA | Request Quote |
| Dr. Reddy’s | 100mg / 400mg | 850K+ boxes | 100 boxes | WHO-GMP, USFDA | Request Quote |
Price & Supply Information (2026 Q2 Latest Quotation)
Wholesale Price Range: USD 11.4–38/120 tablets per box depending on strength, manufacturer, and destination market.
Supply Status: Stable global availability with strong export capacity from India.
Lead Time: 7–15 working days for standard dispatch; urgent procurement may be faster depending on stock.
Download Latest Imatinib Quotation
Clinical Data & FAQ
Key Clinical Study Summary
The IRIS trial confirmed long-term survival advantages for newly diagnosed CML patients treated with Imatinib compared to interferon-based therapy. More than 80% of patients maintained progression-free survival over extended follow-up periods, supporting its role as first-line therapy.
FAQ
1. What are the most common Imatinib side effects?
Common side effects include nausea, vomiting, muscle cramps, swelling around the eyes, fatigue, rash, diarrhea, and mild liver enzyme changes. Most patients can manage these effects with monitoring and supportive care.
2. Is Imatinib safe for long-term use?
Yes. Many CML patients remain on Imatinib for years. Regular blood tests, liver monitoring, and physician supervision are essential to maintain long-term safety and treatment success.
3. Can Imatinib cause serious liver damage?
Although uncommon, hepatotoxicity can occur. Liver function tests should be performed regularly. Immediate medical review is needed if jaundice, severe fatigue, or abdominal pain develops.
4. Is generic Imatinib as effective as Glivec?
Yes. Approved generics manufactured under WHO-GMP and USFDA standards are considered therapeutically equivalent and widely used in international oncology practice.
5. Can Imatinib affect heart health?
Rarely, fluid retention and cardiac complications may occur, especially in older patients or those with existing heart disease. Monitoring is recommended during treatment.
6. Is pregnancy safe during Imatinib therapy?
Imatinib is generally not recommended during pregnancy due to fetal risk. Treatment decisions should always be made with oncology and obstetric specialists.
7. How should Imatinib be taken?
It is usually taken with food and a large glass of water to reduce stomach irritation. The exact dose depends on diagnosis and physician guidance.
8. Where can international buyers source reliable generic Imatinib?
Trusted sourcing partners like 9S Pharmacy Service help buyers access verified manufacturers with regulatory documentation, quotations, and export support.
Related Products & Internal Links
- Dasatinib for CML Treatment
- Nilotinib Safety and Supply Guide
- Lenalidomide Uses in Multiple Myeloma
- Osimertinib for Lung Cancer Survival Rate
- Cheap Cancer Drugs in India Guide
Bottom Related Long-Tail Search List
- best generic Imatinib suppliers in India
- Imatinib side effect and safety for CML patients
- Glivec vs generic Imatinib comparison
- Imatinib wholesale price 2026
- WHO GMP Imatinib manufacturers list
Footer CTA & Trust Signals
Need verified Imatinib supply support?
Contact 9S Pharmacy Service Now
Trust Badges
- WHO-GMP Certified
- USFDA Approved Facilities
- EDQM Compliance Support
- Global Export Documentation
- Verified Manufacturer Network
Contact Email: contact@984online.com
