Sunitinib vs Sorafenib: Key Differences, Uses, Side Effects, Price & Targeted Therapy Guide
When facing advanced cancers such as renal cell carcinoma (RCC) or other solid tumors, patients and oncologists often discuss options like sunitinib and sorafenib. Both are oral multi-targeted tyrosine kinase inhibitors (TKIs) that interfere with cancer cell growth and the formation of new blood vessels (angiogenesis). Yet they differ in their approved uses, specific targets, dosing schedules, and side effect profiles.
This article provides a general, educational comparison of **sunitinib vs sorafenib** to help readers understand their roles in oncology. This content is for informational purposes only and does not replace professional medical advice, diagnosis, or treatment. Treatment decisions must be made by a qualified oncologist based on individual patient factors.
What Are Sunitinib and Sorafenib?

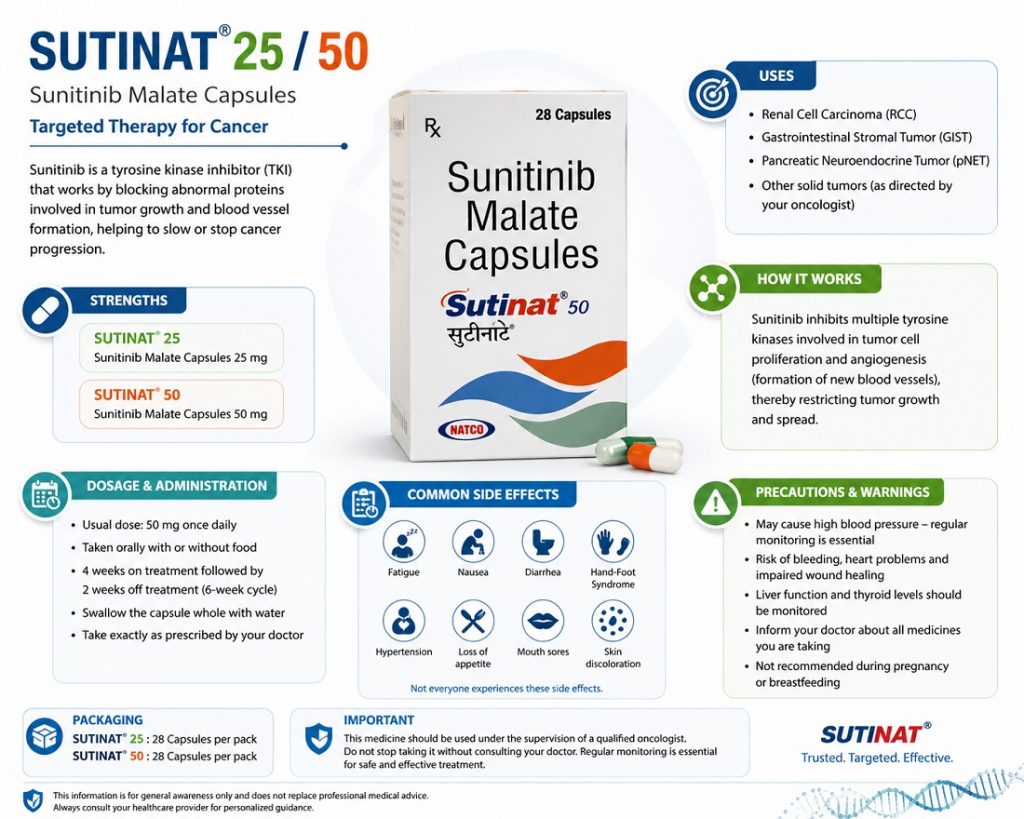
Sunitinib is a multi-targeted tyrosine kinase inhibitor primarily used in:
- Advanced Renal Cell Carcinoma (RCC)
- Gastrointestinal Stromal Tumors (GIST)
- Pancreatic Neuroendocrine Tumors (pNET)
- Selected resistant thyroid cancers
It works by blocking several growth-promoting pathways, especially:
- VEGFR (Vascular Endothelial Growth Factor Receptor)
- PDGFR (Platelet-Derived Growth Factor Receptor)
- KIT
- FLT3
- RET
This helps reduce tumor blood supply and slows cancer progression.
Common brand example: Sutent
Sorafenib is another oral targeted TKI commonly used for:
- Hepatocellular Carcinoma (HCC / Liver Cancer)
- Advanced Renal Cell Carcinoma (RCC)
- Radioactive iodine-resistant thyroid cancer
It inhibits multiple cancer signaling pathways including:
- RAF kinase
- VEGFR
- PDGFR
- FLT3
- RET
This makes it particularly effective in liver cancer where angiogenesis control is critical.
Common brand example: Nexavar
Sunitinib primarily targets vascular endothelial growth factor receptors (VEGFRs), platelet-derived growth factor receptors (PDGFRs), and other kinases. Sorafenib also inhibits VEGFR and PDGFR but additionally targets the RAF/MEK/ERK pathway, which can influence cell proliferation.
These differences in kinase inhibition contribute to their distinct clinical profiles and approved indications.
Approved Indications: Sunitinib Uses vs Sorafenib Uses
Sunitinib (Sutent) Indications
Sunitinib is indicated for:
- Gastrointestinal stromal tumor (GIST) in adults after disease progression on or intolerance to imatinib.
- Advanced renal cell carcinoma (RCC).
- Adjuvant treatment of adult patients at high risk of recurrent RCC following nephrectomy (kidney removal).
- Progressive, well-differentiated pancreatic neuroendocrine tumors (pNET) in adults with unresectable locally advanced or metastatic disease.
Sorafenib (Nexavar) Indications
Sorafenib is indicated for:
- Unresectable hepatocellular carcinoma (HCC, a type of liver cancer).
- Advanced renal cell carcinoma (RCC).
- Locally recurrent or metastatic, progressive differentiated thyroid carcinoma (DTC) that is refractory to radioactive iodine treatment.
Both drugs have established roles in advanced RCC, but sorafenib has a primary indication in HCC, while sunitinib has broader use in GIST and pNET. Overlap exists in kidney cancer, where both have been studied as options, though treatment landscapes have evolved with newer therapies and combinations.

Mechanism of Action: How Sunitinib and Sorafenib Work
Both medications disrupt tumor-supporting pathways, but with nuances:
- Sunitinib strongly inhibits VEGFR-1, -2, -3 and PDGFR, affecting angiogenesis and tumor cell signaling. It also impacts other receptors like KIT and FLT3.
- Sorafenib inhibits VEGFR and PDGFR similarly but adds inhibition of RAF kinase, potentially affecting the MAP kinase pathway involved in cell growth and survival.
These mechanisms make both drugs “anti-angiogenic,” limiting the tumor’s blood supply, while also directly affecting cancer cell behavior in responsive tumors.
Dosing and Administration
Sunitinib is typically dosed at 50 mg once daily for 4 weeks on treatment followed by 2 weeks off (4/2 schedule) for GIST and advanced RCC. For pNET, a continuous 37.5 mg daily dose without scheduled breaks is often used. Doses may be adjusted in 12.5 mg increments based on tolerability.
Sorafenib is usually taken as 400 mg twice daily (800 mg total per day) continuously, with or without food, though dose reductions or interruptions are common for side effects such as hand-foot skin reactions.
Patients should take these medications exactly as prescribed and report any difficulties with adherence promptly.

Side Effects Comparison: Sunitinib vs Sorafenib
Both drugs share several common side effects typical of VEGF pathway inhibitors, including fatigue, diarrhea, hypertension (high blood pressure), and hand-foot skin reactions. However, differences in frequency and severity exist.
Common side effects with **sunitinib** often include:
- Fatigue/asthenia
- Diarrhea, nausea, vomiting, stomatitis (mouth sores)
- Hand-foot syndrome
- Hypertension
- Skin/hair color changes, altered taste
- Decreased appetite
Common side effects with **sorafenib** frequently include:
- Hand-foot skin reactions (often prominent)
- Diarrhea
- Fatigue
- Rash
- Hypertension
- Weight loss, loss of appetite
- Hair thinning or changes
Some studies suggest sorafenib may have a lower overall incidence of certain severe (grade 3/4) hematologic toxicities compared to sunitinib in specific settings like first-line metastatic RCC, while sunitinib may show higher rates of neutropenia, thrombocytopenia, or stomatitis. Hand-foot reactions can be particularly bothersome with sorafenib. Both can cause liver enzyme elevations and require monitoring; sunitinib has a boxed warning for hepatotoxicity.
Other serious risks for both include bleeding events, cardiac effects, thyroid dysfunction, and impaired wound healing. Regular blood pressure monitoring, liver function tests, and complete blood counts are essential during treatment.
Important: Side effect experiences vary widely between patients. Proactive management with supportive care, dose adjustments, or temporary pauses can help many individuals continue therapy.
Key Differences: Sunitinib vs Sorafenib Summary Table
While direct head-to-head data can vary by cancer type and line of therapy, general distinctions include:
- Primary strengths: Sorafenib has a well-established role in unresectable HCC. Sunitinib offers options in GIST (post-imatinib), pNET, and adjuvant RCC.
- RCC context: Both have been used in advanced RCC; some analyses indicate comparable efficacy in certain populations with differences in toxicity profiles.
- Toxicity patterns: Sorafenib often linked to more pronounced skin reactions; sunitinib may have more hematologic or mucosal effects in some patients.
- Dosing convenience: Sunitinib uses intermittent scheduling in many indications; sorafenib is typically continuous daily dosing.
Modern treatment algorithms for RCC frequently incorporate immunotherapy combinations or other TKIs, so the role of these older agents continues to evolve based on guidelines and patient-specific factors.
Choosing Between Sunitinib and Sorafenib: Factors to Consider
Selection depends on the exact cancer type and stage, prior treatments, comorbidities (e.g., liver or heart function), patient performance status, and potential drug interactions. Cost, availability, and insurance coverage may also influence decisions in different healthcare systems.
Oncologists weigh the potential benefits against risks for each individual. No single drug is universally “better”—the goal is finding the most appropriate option for the patient’s situation at that time.
FAQs About Sunitinib vs Sorafenib
What is the main difference between sunitinib and sorafenib?
Sunitinib and sorafenib are both multi-kinase inhibitors, but sorafenib additionally targets the RAF pathway and is primarily approved for unresectable hepatocellular carcinoma (liver cancer), while sunitinib has approvals in GIST, pancreatic neuroendocrine tumors, and adjuvant RCC. Their side effect profiles also differ slightly in frequency of certain toxicities.
Are sunitinib and sorafenib used for the same cancer?
They overlap in advanced renal cell carcinoma (kidney cancer). Sorafenib is notably used for liver cancer (HCC), and sunitinib for GIST and pNET. Choice depends on the specific diagnosis and treatment line.
Which has more side effects: sunitinib or sorafenib?
Both can cause significant side effects. Some analyses suggest sorafenib may have lower rates of certain severe hematologic toxicities, while sunitinib can be associated with higher rates of fatigue, stomatitis, or blood count changes in specific settings. Individual tolerance varies greatly.
Can sunitinib and sorafenib be taken together?
They are not typically combined due to overlapping mechanisms and increased toxicity risk. Sequential use (one after the other) has been studied in some RCC cases, but this must be decided by the treating physician.
Is one drug better than the other for kidney cancer?
In advanced RCC, both have shown activity. Comparative studies show mixed results regarding efficacy and toxicity, with no clear universal winner. Newer combination therapies have changed first-line standards in many cases.
Do sunitinib and sorafenib cause hand-foot syndrome?
Yes, hand-foot skin reactions are common with both, though often more frequent or severe with sorafenib. Preventive skin care, moisturizers, and dose adjustments can help manage this side effect.
How long do patients usually take sunitinib or sorafenib?
Treatment duration varies. It continues as long as there is clinical benefit and acceptable tolerability, or for a planned duration in adjuvant settings. Regular scans and assessments guide continuation or discontinuation.
What monitoring is needed with these medications?
Regular blood pressure checks, liver and thyroid function tests, complete blood counts, and cardiac monitoring when indicated. Patients should report symptoms like severe fatigue, bleeding, chest pain, or yellowing of the skin immediately.
Are sunitinib and sorafenib chemotherapy?
No. They are targeted therapies (tyrosine kinase inhibitors), not traditional cytotoxic chemotherapy. They specifically block enzymes involved in cancer signaling rather than broadly killing dividing cells.
Can these drugs be used in patients with liver problems?
Both require caution in patients with liver impairment. Sorafenib is used in HCC (liver cancer), but dosing and monitoring are critical. Sunitinib carries a boxed warning for hepatotoxicity. Liver function must be carefully assessed.
What should I do if I experience severe side effects?
Contact your oncology team immediately. Many side effects can be managed with supportive medications, dose reductions, or temporary treatment breaks. Never stop or adjust the dose without medical guidance.
Are there newer alternatives to sunitinib and sorafenib?
Yes. Treatment for RCC, HCC, and other cancers has advanced with immunotherapy-TKI combinations and next-generation agents. Discuss current guideline-recommended options with your doctor.
Conclusion
Sunitinib and sorafenib represent important milestones in targeted cancer therapy. While they share anti-angiogenic properties, their approved indications, additional kinase targets, dosing regimens, and tolerability profiles differ in meaningful ways. Sunitinib offers options in GIST, pNET, and RCC (including adjuvant), whereas sorafenib is a recognized choice for unresectable HCC and certain thyroid cancers, with established use in RCC.
Ultimately, the choice between sunitinib vs sorafenib—or any other therapy—depends on many individual factors. Open communication with your oncology care team is essential to weigh benefits, risks, and quality-of-life considerations.
If you have questions about these medications or your treatment plan, speak directly with your healthcare provider for personalized guidance. Staying informed while relying on expert medical advice supports the best possible care.
Disclaimer: This article is educational and based on general information regarding approved uses of sunitinib and sorafenib. It does not constitute medical advice. Always consult your physician before starting, stopping, or changing any treatment. Individual results and experiences vary.

