Imatinib India Generic Manufacturer India
Imatinib India Generic | Trusted Supply & Pricing 2026
Meta Description
Source Imatinib India Generic from WHO-GMP certified manufacturers in India. Compare prices, clinical data, and wholesale procurement options for 2026.
2. Hero Section (Above-the-Fold)
Hero Subtitle
Access high-quality Imatinib India Generic from trusted Indian manufacturers with WHO-GMP, USFDA, and EU-GMP compliance. Ideal for hospitals, distributors, procurement teams, and patients seeking affordable long-term oncology treatment solutions.
CTA Buttons
Suggested Hero Image Alt Text
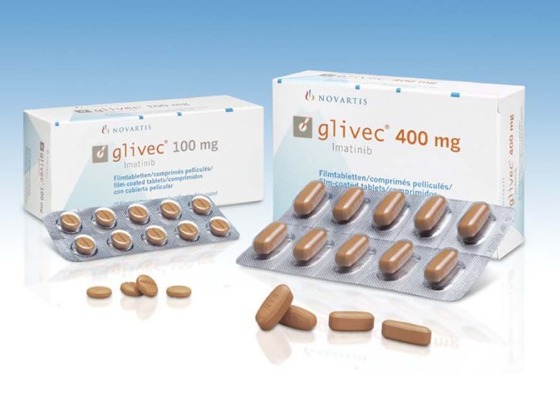
3. Drug Overview
Imatinib is a targeted anti-cancer medicine classified as a tyrosine kinase inhibitor (TKI). It is widely used for the treatment of chronic myeloid leukemia (CML), Philadelphia chromosome-positive acute lymphoblastic leukemia (Ph+ ALL), gastrointestinal stromal tumors (GIST), dermatofibrosarcoma protuberans, and selected myelodysplastic and eosinophilic disorders.
The original innovator brand is Gleevec®/Glivec® developed by Novartis. It became one of the most important breakthroughs in oncology because it changed CML from a life-threatening disease into a manageable long-term condition for many patients.
Imatinib works by blocking abnormal tyrosine kinases including BCR-ABL, KIT, and PDGFR. In CML, the BCR-ABL fusion protein causes uncontrolled white blood cell growth. By inhibiting this pathway, Imatinib helps stop cancer progression and improves long-term survival.
FDA information confirms Imatinib is approved for CML and related indications. The landmark IRIS study showed strong long-term outcomes, with approximately 83.3% overall survival at 10 years for first-line therapy patients, helping establish Imatinib as the global standard of care. Sources: FDA Gleevec information and IRIS long-term study follow-up.
After patent expiry, India became a major center for Imatinib India Generic manufacturing. Companies such as Cipla, Natco Pharma, Hetero, Sun Pharma, and Dr. Reddy’s provide high-quality generic Imatinib for domestic and export markets. These products offer the same active ingredient and therapeutic effect at significantly lower prices—often reducing treatment cost by 70–90% compared to innovator pricing.
Original Brand vs India Generic
| Parameter | Original Brand | Imatinib India Generic |
|---|---|---|
| Brand | Gleevec / Glivec | Generic Imatinib |
| Active Ingredient | Imatinib Mesylate | Imatinib Mesylate |
| Strengths | 100mg / 400mg | 100mg / 400mg |
| Manufacturer | Novartis | Cipla / Natco / Hetero / Sun Pharma |
| Cost | Premium | 70–90% lower |
| Export Supply | Limited by pricing | High-volume global supply |
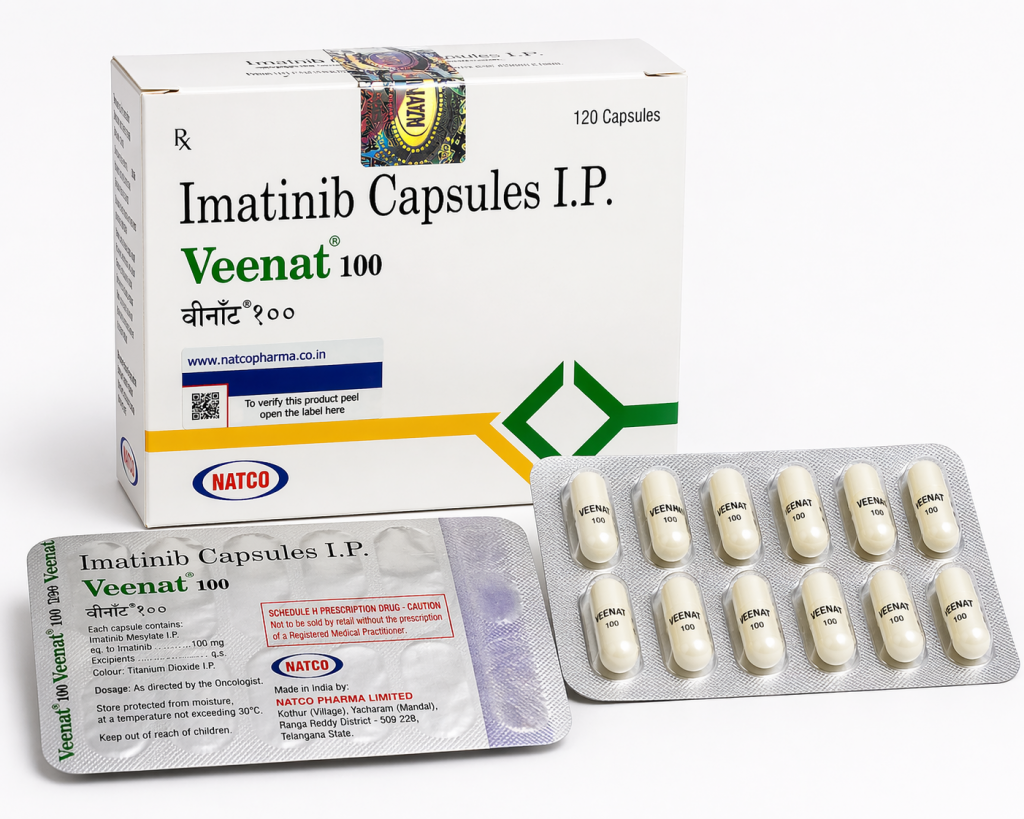
4. Why Choose India Generic
- Major Cost Savings: Imatinib India Generic commonly reduces procurement cost by 70–90% compared with innovator pricing.
- WHO-GMP Certified Facilities: Leading manufacturers maintain WHO-GMP, USFDA, EU-GMP, and export-compliant facilities.
- Large Production Capacity: Indian pharma leaders support large monthly output for hospital tenders and distributor networks.
- Strong Oncology Expertise: Specialized oncology divisions ensure quality handling and regulatory documentation.
- Flexible MOQ: Hospitals, NGOs, wholesalers, and private importers can source based on practical order volumes.
- 9S Network Assurance: Verified supplier screening reduces counterfeit risk and improves procurement confidence.
- Stable Export Documentation: COA, COPP, batch records, and stability reports are available for global registration support.
WHO market reports continue to identify India as one of the world’s largest suppliers of generic medicines, especially in oncology and chronic therapy categories, making Imatinib India Generic a dependable long-term sourcing option.
5. Member Manufacturers Directory
| Company | Strengths Available | Monthly Capacity | MOQ | Certification | Action |
|---|---|---|---|---|---|
| Cipla | 100mg / 400mg | 2M+ tablets | 500 boxes | WHO-GMP, USFDA | Request Quote |
| Natco Pharma | 100mg / 400mg | 1.5M+ tablets | 300 boxes | WHO-GMP, EU-GMP | Request Quote |
| Hetero | 100mg / 400mg | 2M+ tablets | 500 boxes | WHO-GMP, USFDA | Request Quote |
| Sun Pharma | 100mg / 400mg | 2.5M+ tablets | 500 boxes | WHO-GMP, USFDA | Request Quote |
| Dr. Reddy’s | 100mg / 400mg | 1.8M+ tablets | 400 boxes | WHO-GMP, USFDA | Request Quote |
| Zydus | 100mg / 400mg | 1.2M+ tablets | 300 boxes | WHO-GMP | Request Quote |
| Intas | 100mg / 400mg | 1M+ tablets | 300 boxes | WHO-GMP, EU-GMP | Request Quote |
| Glenmark | 100mg / 400mg | 900K+ tablets | 300 boxes | WHO-GMP | Request Quote |
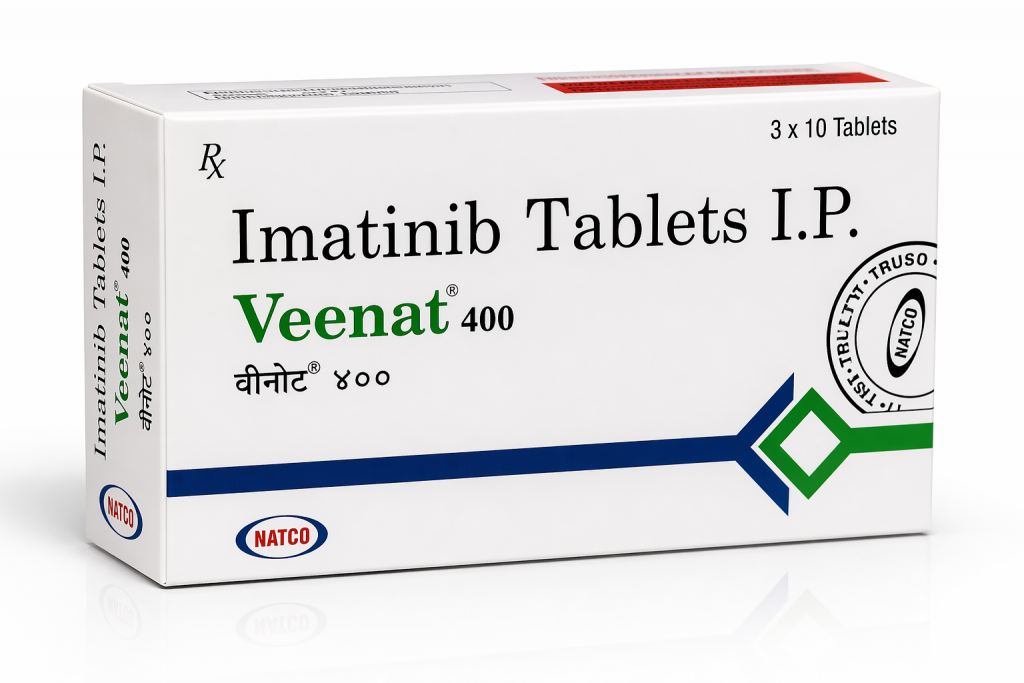
6. Price & Supply Information (2026 Q2 Latest Quotation)
Imatinib 100mg: USD 11.5– 36/120 tablets per box
Imatinib 400mg: USD 34–38 /120 tablet per box
Final pricing depends on destination country, registration requirements, shipping method, packaging customization, and order quantity.
Supply Status: Stable
Lead Time: 2–4 weeks
Urgent dispatch available for repeat buyers and institutional orders.
Download Latest Imatinib Quotation
7. Clinical Data & FAQ
Clinical Summary
The IRIS trial confirmed Imatinib as the first-line standard treatment for newly diagnosed CML. Long-term follow-up showed strong molecular response, durable remission, and estimated 10-year overall survival above 80%. This remains one of the most important clinical success stories in targeted oncology therapy.
FAQ
Q1. What is Imatinib used for?
Imatinib is mainly used for CML, GIST, and several rare blood disorders. It blocks abnormal proteins that drive cancer growth and helps patients maintain long-term disease control.
Q2. Is Imatinib India Generic effective?
Yes. When sourced from WHO-GMP or USFDA-compliant manufacturers, Imatinib India Generic provides the same active ingredient and expected therapeutic outcome as the original brand.
Q3. Why is Indian generic Imatinib cheaper?
Large-scale production, strong generic competition, and API integration reduce cost while maintaining international quality standards.
Q4. Can hospitals procure directly from India?
Yes. Hospitals, distributors, NGOs, and procurement agencies commonly source directly with export documents and regulatory support.
Q5. What strengths are commonly available?
The most common strengths are 100mg and 400mg tablets used across major oncology protocols.
Q6. Is prescription required?
Yes. Imatinib is a prescription-only anti-cancer medicine and should be used only under physician supervision.
Q7. What certifications should buyers verify?
WHO-GMP, USFDA, EU-GMP, COA, and manufacturing license verification are strongly recommended.
Q8. What is the MOQ for export orders?
Most suppliers offer starting quantities between 300 and 500 boxes depending on destination and registration requirements.
8. Related Products & Internal Links
- Dasatinib Generic India
- Nilotinib India Generic
- Erlotinib Generic Supplier
- Lenalidomide Manufacturer India
- Sorafenib Generic Export India
Related Long-Tail Searches:
best Imatinib India Generic supplier 2026
WHO GMP Imatinib manufacturer India
wholesale Imatinib 400mg generic India
buy Imatinib generic from India
Imatinib oncology supplier export India
9. Footer CTA & Trust Signals
Access trusted oncology sourcing through verified Indian manufacturers with global export capability.
Request Your 2026 Imatinib Quote Now
Trust Badges
- WHO-GMP Certified
- USFDA Approved Export Facilities
- EU-GMP Compliance
- EDQM Documentation Support
- Global Tender Procurement Experience
Contact
Email: contact@984online.com
Trusted by procurement professionals, hospitals, oncology distributors, and patients seeking safe access to Imatinib India Generic in 2026.
