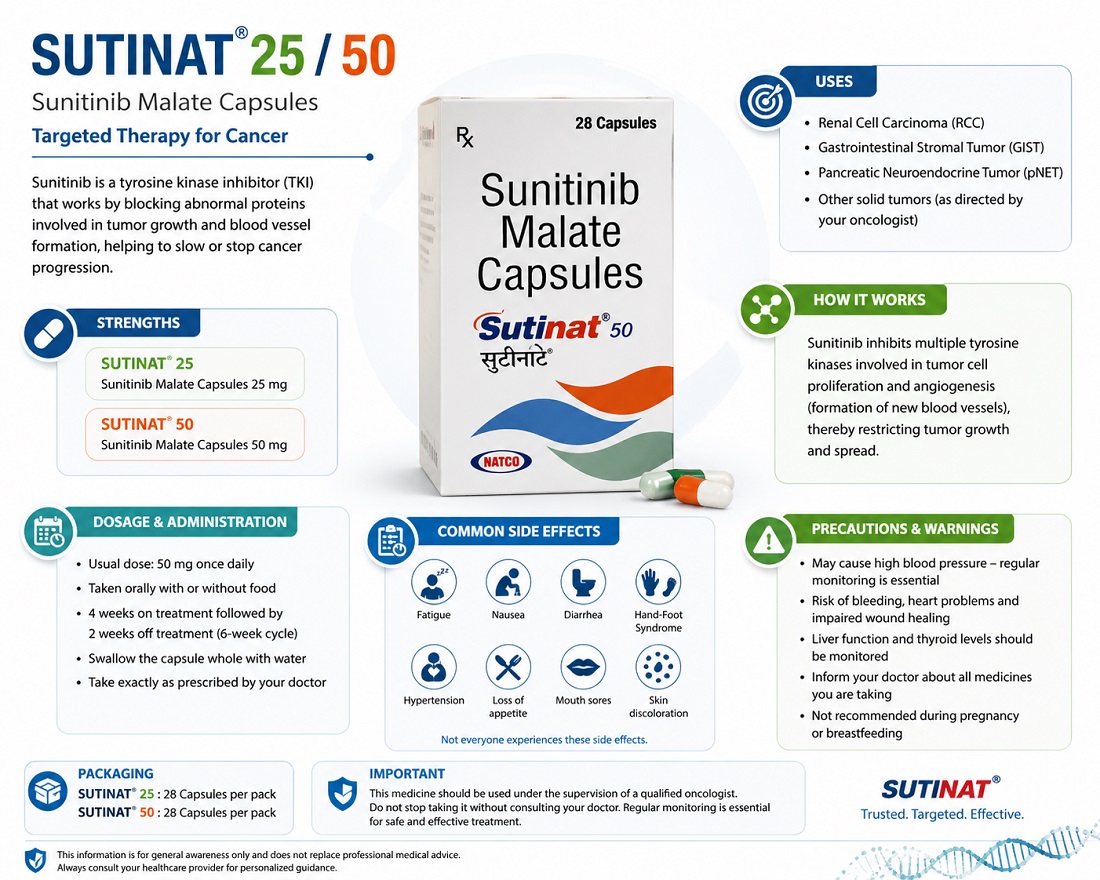
Sunitinib Uses: Treatment for Advanced Kidney Cancer, GIST and Pancreatic Tumors
Sunitinib is an important targeted cancer medicine used in the treatment of several advanced cancers, especially renal cell carcinoma (kidney cancer), gastrointestinal stromal tumors (GIST), and pancreatic neuroendocrine tumors (pNET). It belongs to a class of medicines called multi-targeted tyrosine kinase inhibitors (TKIs), designed to block cancer growth pathways and reduce tumor blood supply.
Unlike traditional chemotherapy, sunitinib works by targeting specific proteins involved in tumor growth and angiogenesis. This makes it a major treatment option for patients with advanced or resistant cancers where surgery or standard chemotherapy may no longer be sufficient.
Because sunitinib is a high-risk prescription oncology medicine, treatment must always be supervised by an oncologist. Proper dosage, monitoring, and safe sourcing are essential for both effectiveness and patient safety.
What Is Sunitinib?
Sunitinib is an oral multi-targeted receptor tyrosine kinase inhibitor. It works by blocking several enzymes (kinases) involved in tumor growth and angiogenesis—the process by which tumors develop their own blood supply. By inhibiting pathways such as vascular endothelial growth factor receptor (VEGFR) and platelet-derived growth factor receptor (PDGFR), among others, sunitinib can slow or stop the progression of certain cancers.
It is typically taken in cycles, often 4 weeks on treatment followed by 2 weeks off, though dosing may vary based on the specific condition and individual patient factors. Sunitinib is available in capsule form in different strengths.

Approved Uses of Sunitinib (Sutent)
Sunitinib has specific approved indications for adult patients with certain cancers. Its uses focus on situations where the disease is advanced, progressive, or has not responded adequately to prior treatments.
Gastrointestinal Stromal Tumor (GIST)
Sunitinib is indicated for the treatment of gastrointestinal stromal tumor (GIST) in adult patients after disease progression on or intolerance to imatinib mesylate (commonly known as Gleevec). GIST is a rare type of tumor that usually arises in the wall of the stomach or intestines. When imatinib is no longer effective or cannot be tolerated, sunitinib may be considered as a subsequent targeted option.
Advanced Renal Cell Carcinoma (RCC)
One of the most established sunitinib uses is in advanced renal cell carcinoma (kidney cancer) that has spread or cannot be removed surgically. It is used in patients with advanced RCC as a treatment to help manage the disease.
Sunitinib is also approved for the adjuvant treatment of adult patients at high risk of recurrent RCC following nephrectomy (surgical removal of the kidney). In this setting, it is used to help reduce the risk of the cancer returning after surgery in high-risk cases.
Pancreatic Neuroendocrine Tumors (pNET)
Sunitinib is indicated for the treatment of progressive, well-differentiated pancreatic neuroendocrine tumors (pNET) in adult patients with unresectable locally advanced or metastatic disease. These are tumors of the pancreas that produce hormones and have progressed despite other approaches or cannot be surgically removed.
These are the primary approved uses based on regulatory approvals. Sunitinib is not intended for all cancer types and should only be used under the supervision of an oncologist experienced in managing these conditions.
How Does Sunitinib Work? Mechanism of Action
As a multi-kinase inhibitor, sunitinib targets multiple receptors on cell surfaces that play roles in cancer cell proliferation and the formation of new blood vessels that feed tumors. By blocking these signals, it can lead to reduced tumor growth and, in some cases, tumor shrinkage or stabilization.
This targeted approach differs from traditional chemotherapy, which affects rapidly dividing cells more broadly. However, because the targeted pathways are also present in some healthy tissues, side effects can still occur.
General Information on Dosing and Administration
Sunitinib is taken orally, usually once daily, with or without food. A common schedule for GIST and advanced RCC is 50 mg daily for 4 weeks, followed by a 2-week break (Schedule 4/2). For adjuvant RCC treatment, the same schedule may be used for a defined number of cycles. For pNET, dosing may differ and is often continuous without the scheduled break.
Doses can be adjusted by the doctor in 12.5 mg increments based on how well the patient tolerates the medication and any side effects experienced. Treatment continues as long as there is clinical benefit and the medication remains tolerable, or until disease progression or unacceptable toxicity occurs.
Patients are advised to follow their healthcare provider’s instructions precisely and report any new symptoms promptly.
Important Safety Information and Potential Side Effects
Like all medications, sunitinib can cause side effects. Common adverse reactions reported in clinical use (occurring in 25% or more of patients in studies) include fatigue/asthenia, diarrhea, mucositis/stomatitis (mouth sores or inflammation), nausea, decreased appetite, vomiting, abdominal pain, hand-foot syndrome (redness, swelling, or pain on palms and soles), hypertension (high blood pressure), bleeding events, altered taste, and dyspepsia.
Other possible effects include skin discoloration (sometimes yellowing due to the drug), hair color changes, dry skin, and swelling. Serious risks may include severe liver problems (hepatotoxicity), heart issues, bleeding, gastrointestinal perforation (rare), thyroid dysfunction, and wound healing complications.
Sunitinib carries a boxed warning for hepatotoxicity, and patients should be monitored regularly with blood tests. It can cause fetal harm and is not recommended during pregnancy. Effective contraception is advised for those who can become pregnant during treatment and for a period after.
This information is general and educational. Individual side effects and risks vary. Patients must discuss all potential risks and benefits with their doctor and report symptoms such as severe fatigue, unusual bleeding, yellowing of the skin/eyes, severe diarrhea, or chest pain immediately. Regular monitoring, including blood pressure checks, liver function tests, and cardiac evaluation when appropriate, is essential during treatment.
Living with Sunitinib Treatment: Practical Considerations
Patients on sunitinib often work closely with their oncology team to manage side effects proactively. Strategies may include supportive medications for nausea or diarrhea, skin care routines for hand-foot syndrome, dietary adjustments, and blood pressure management.
Because treatment can affect energy levels, taste, and appetite, maintaining nutrition and hydration is important. Lifestyle factors, such as avoiding grapefruit juice (which may interact with the drug) and informing the doctor about all other medications or supplements, help optimize safety.
Treatment decisions are highly individualized, taking into account the specific cancer type, stage, prior therapies, overall health, and patient preferences.
FAQs About Sunitinib Uses
What is sunitinib primarily used for?
Sunitinib is primarily used as a targeted therapy for specific advanced cancers, including gastrointestinal stromal tumor (GIST) after imatinib failure or intolerance, advanced renal cell carcinoma (kidney cancer), adjuvant treatment in high-risk RCC after kidney surgery, and progressive well-differentiated pancreatic neuroendocrine tumors (pNET) that are unresectable or metastatic. It helps interfere with cancer cell growth signals and blood vessel formation in tumors.
Is sunitinib used for kidney cancer?
Yes, sunitinib is approved for the treatment of advanced renal cell carcinoma (RCC) and as adjuvant therapy in patients at high risk of recurrence after surgical removal of the kidney. It is one of the established options in the management of certain kidney cancers.
How does sunitinib work in cancer treatment?
Sunitinib inhibits multiple receptor tyrosine kinases involved in tumor proliferation and angiogenesis. By blocking these pathways, it can slow cancer growth and limit the tumor’s ability to develop a blood supply, potentially leading to disease stabilization or response in responsive patients.
What are the most common side effects of sunitinib?
Common side effects include fatigue, diarrhea, mouth sores or inflammation, nausea, vomiting, decreased appetite, hand-foot syndrome, high blood pressure, altered taste, and skin or hair changes. Many side effects are manageable with supportive care, dose adjustments, or temporary interruptions.
Can sunitinib be taken with food?
Yes, sunitinib capsules can generally be taken with or without food. Patients should follow their prescribing doctor’s specific instructions and maintain consistency in how they take it.
Is sunitinib a chemotherapy drug?
No, sunitinib is not traditional chemotherapy. It is a targeted therapy classified as a tyrosine kinase inhibitor. It specifically blocks certain proteins involved in cancer growth rather than broadly affecting all rapidly dividing cells.
How long do patients typically take sunitinib?
Treatment duration varies. It may continue for as long as the patient derives clinical benefit and tolerates the medication, or for a predefined number of cycles in the adjuvant setting. Regular assessments determine whether to continue, adjust, or stop therapy.
Does sunitinib cause hair loss?
Hair color changes or thinning can occur, and some patients experience hair depigmentation or loss. These effects are usually reversible after stopping treatment, but patients should discuss any concerns with their doctor.
What monitoring is required during sunitinib treatment?
Regular blood tests for liver function, complete blood counts, thyroid function, and blood pressure monitoring are typically required. Cardiac evaluation may be needed in some cases. Patients should attend all scheduled follow-up appointments.
Can sunitinib be used in combination with other treatments?
In approved indications, sunitinib is often used as monotherapy. Combination use depends on the specific cancer, treatment guidelines, and ongoing clinical research. Any combination therapy must be decided by the treating oncologist.
Is sunitinib safe during pregnancy?
No. Based on its mechanism and animal studies, sunitinib can cause fetal harm. It is contraindicated in pregnancy. Patients who can become pregnant should use effective contraception during treatment and for an appropriate period afterward, as advised by their doctor.
What should I do if I miss a dose of sunitinib?
Patients should contact their healthcare provider or pharmacist for guidance on missed doses. Do not double the dose to make up for a missed one unless specifically instructed.
Conclusion
Sunitinib remains an important targeted option in the management of specific advanced cancers, particularly certain cases of renal cell carcinoma, GIST after prior therapy, and progressive pNET. Its ability to inhibit key pathways involved in tumor growth and angiogenesis has made it a valuable tool in oncology when used appropriately.
While effective for indicated uses, treatment requires careful medical supervision due to potential side effects and the need for individualized dosing and monitoring. Decisions about starting or continuing sunitinib should always be made in consultation with a qualified oncologist who can weigh benefits against risks for each patient.
If you or a loved one has questions about sunitinib or cancer treatment options, speak with your healthcare team for the most current and personalized information. Early and open communication with your doctor helps ensure the safest and most effective care possible.
Disclaimer: This article is educational and based on general publicly available information about approved uses. It does not replace professional medical advice, diagnosis, or treatment. Never start, stop, or change any medication without consulting your physician.
